move crosshair over imaged FRAP point (this requires the stage was properly calibrated before beginning the process)
move stage and repeat 5 or more times
save
Miniscanner / FRAP notes About the miniscanner. Link to brochure.
last updated 20190531 1400
For more general info showing FRAP, go here and follow the links on the web page for examples.
Critical safety concern to prevent camera from being damaged: Important to have camera not in live mode when stimulating. |
Click here to skip directly to illustrated example of FRAP/photoactivation/ablation.
Confocal imaging may be done through the violet FRAP filter cube (all colors but 405 ok).
ROI menu by rightclick hold on image.
Line ROIs by using ROI simple editor.
ROIs may be copied by shift-click.
Multiply ROIs chosen by ctrl-click.
Right click on an ROI to delete or other menu. (Probably click on ROI and hit delete or backspace key too. Try things!)
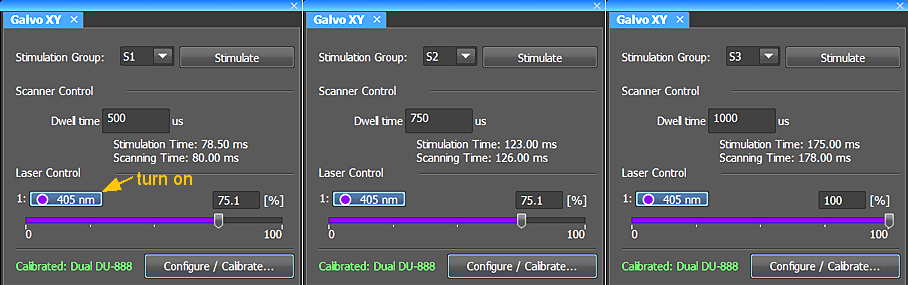
Three stimulation groups. Each group may have different laser intensity and dwell time.
Each group may have multiple ROIs. Define the ROIs and then select them and right click to add to a Stimulation Group.
To FRAP:
FRAP OC button.
Galvo XY tab
Laser power and dwell
Make ROIs
Right click hold on image to get ROI menu or use Simple ROI editor which opens a new window that floats
Shift click on existing ROI to copy it
Click drag on ROI to move it
Control click multiples
Right click, menu add to stim group
Turn off live mode before hitting stimulate.
To include Stim in an experiment,
Application menu, Define sequential stimulation.
screenshot of first training session
In rows, define imaging, stimulation, and wait. Times for stimulation appear to be ignored when shorter than actual stim time.
For instance, Image in one row. Next row stimulation. Next row imaging a timelapse by setting intervals and loops.
Rouds may be grouped and looped.
Ctril click to select rows and [] to group. Put loops and intervals in group column.
Check "close active shutter" to turn off the FRAP laser during imaging.
Check "do time measurement".
If need to switch filter blocks during experiment, need to use ND Sequencer.
To calibrate FRAP:
Galvo XY > calibrate > set button > calibrate button (original version of instructions said to loo in Frap tab/pad)
Turn off imaging lasers
Autoscale
FRAP pad
405 laser approx 3%
camera gain low, low exposure, autocontrast button, make sure LUT histogram shows reflected spot not too bright (e.g. 6000 or lower)
Image H&E or Plexi with Galvo XY tab > configure > calibrate button > set button
In FRAP pad calibrate
move crosshair over imaged FRAP point (this requires the stage was properly calibrated before beginning the process)
move stage and repeat 5 or more times
save

In other accounts, the calibration may be loaded.
The following example exposes three different ROIs each with its own intensity and exposure time. After each exposure, an image is collected.
ROIs (regions of interest) need to be put on images. To access the ROI editor:

Each ROI is in a Stimulation Group with its own intensity and pixel dwell time:
Fifth timepoint shown to illistrate the ROIs and the resultant photconversions and bleaching after four expousres to 405 nm light:
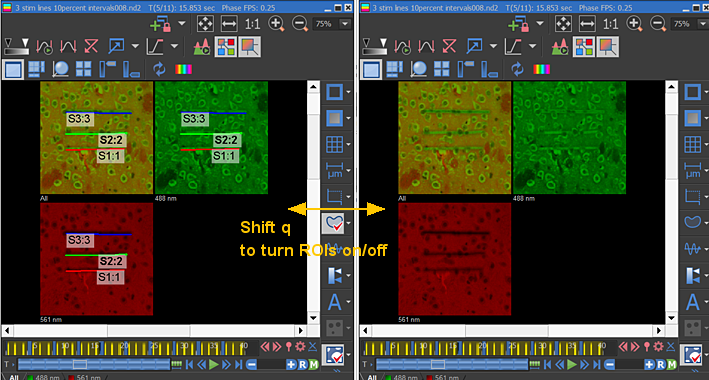
Time sequence of all images:

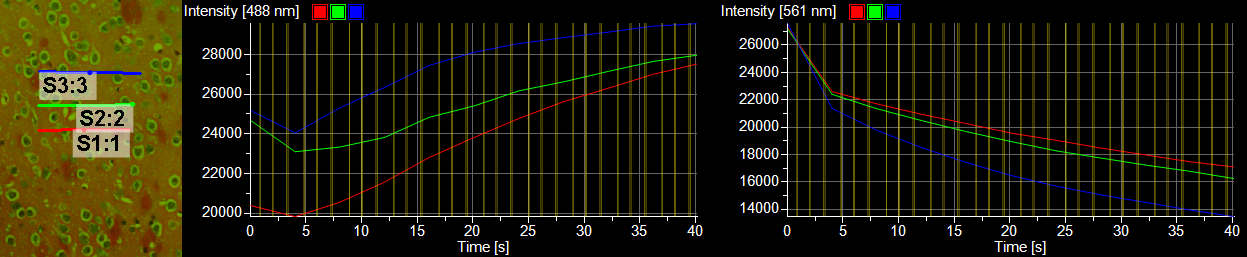
When Perform Time Measurement is checked in the ND Stimulation window, intensity plots through time are generated.
![]()
The grouping pull down menu has an option for mixing stimulation groups instead of having to do them in sequence manually as I set it up. We need to test how this works, however,before relying on it.
About the miniscanner. Link to brochure.
Parts as listed on original purchase order:
77059005 Miniscanner for high speed raster photostimulation. 3mm galvo pair forwavelengths 405-640 nm. Control box included. Scan lens and microscopeadapter not included.
77059007 Scan optics and adapter for Miniscanner to Ti-LAPP system. Replaces Ti-LA-SB sub-branch. Requires main branch.
77016061 Short Gantry Stand 1 99315 Dovetail Mount for Photoactivation Point Scanner.
77097047 LUN-F Laser Combiner with two FC APC single mode output fiber. Fiber 1:405m (50mW); Fiber 2: 473nm (50mW)
77013205 Custom Cable for 6723 DAQ to 16 BNC 1 97337 C-TIRF Filter Cube 405nm Reflection with 320nm and 720nm Transmission
77074319 ZT473rdc, laser reflecting dichroic, R 473nm, T 495-900nm, mounted inTE2000/Ti cube