This paper from 2010 http://www.jneurosci.org/content/20/10/3663.long suggests that biologists should revisit the technique for high resolution imaging in biological tissues and cell culture.
A highly underutilized (in biology) confocal imaging mode is reflectance. When I first used the confocal microscope in 1991, we looked at nickle and silver enhanced staining in cerebellum and lead ATPase labeling for bile canaliculi in liver, however, we (correctly) switched to fluorescence. Reflection continues to be used for looking at cell-substrate interactions, for imaging collagen, and for imaging the surface of materials (including crystals) or assessing the depth of small chambers.
Collection of cell subtrate images on Flickr:
https://www.flickr.com/search/?user_id=39998519%40N00&sort=date-taken-
desc&path=mcammer&nsid=&page=&details=1&tags=irm&view_all=1
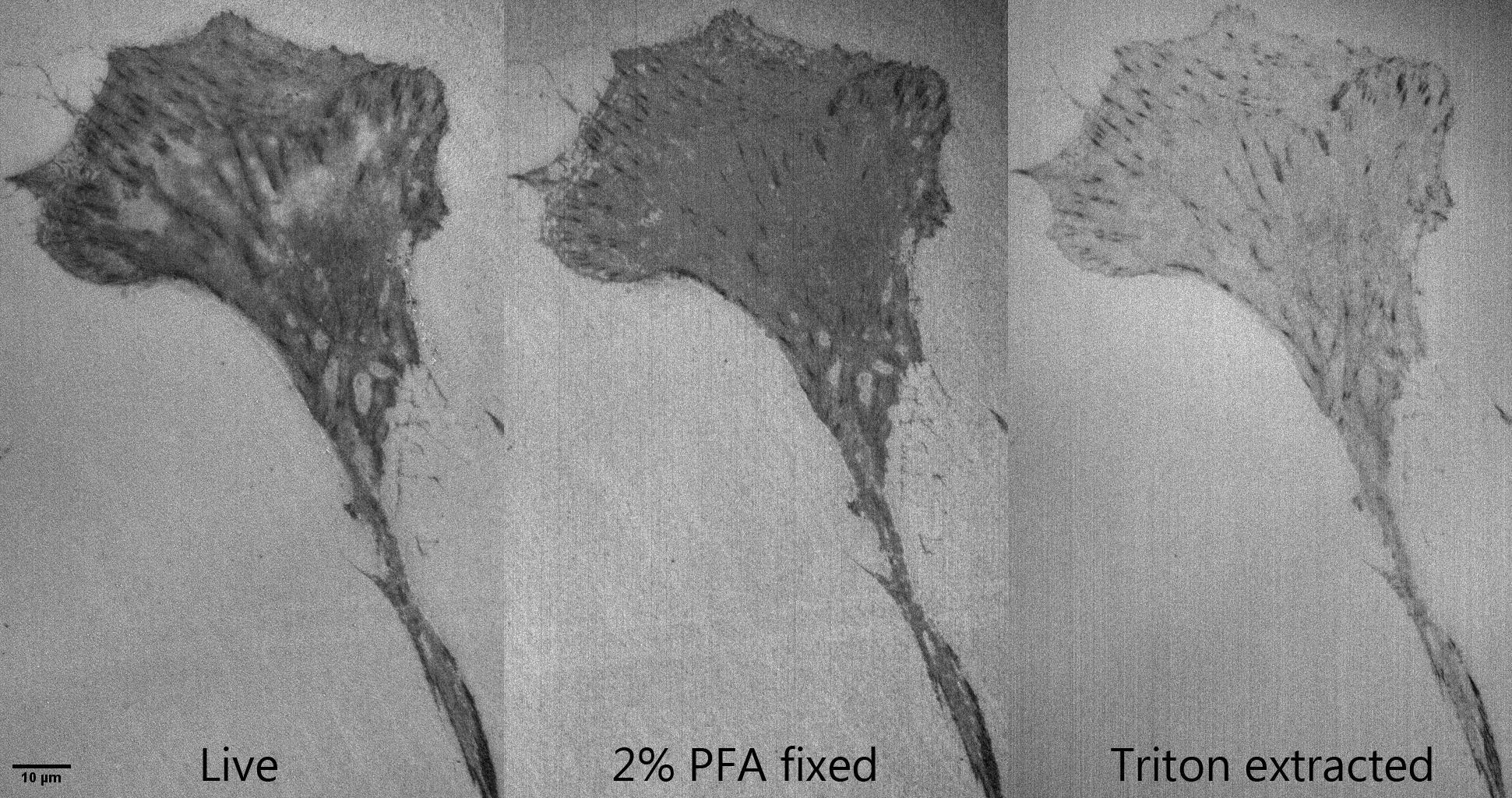
Live vs fixed vs extracted:
https://www.flickr.com/photos/mcammer/27551679825/
Material:
https://www.flickr.com/photos/mcammer/13881401314/
A webpage from early 2000s showing IRM: http://cammer.net/historical/aif/instructions/irm/index.htm
Explanation of an application to measure amounts of cell spreading.
Here's something fun we did in a microscopy class in Aug 2014.
We watched what happened to live cells when they were fixed and then lipid extracted. Goals of the lesson:
- reflection microscopy with confocal to show substrate interaction
- show that fixation alters morphology
- show that extraction further alters morphology and removes material from cells
- staining step shows fast tight binding of phalloidin that saturates faster than a confocal scan (2 ul Alexa 488 phalloidin in 1 ml PBS)
- staining a contrast mechanism to show specific protein -- discussed how f-actin is not washed out by lipid extraction but other proteins may be

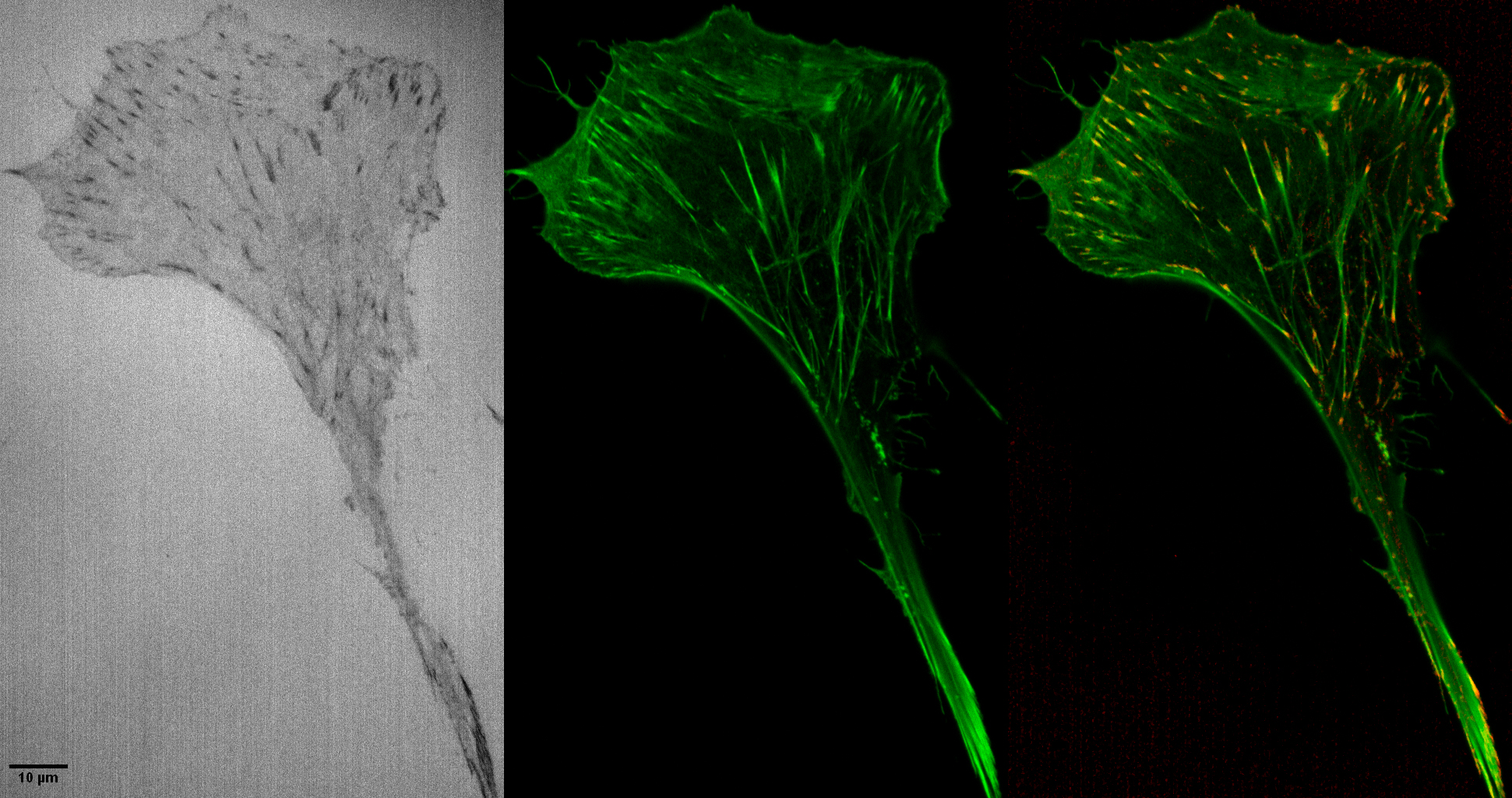
The red image at the right looks like staining for focal adhesion proteins, such as paxillin, but is not. The red dots are the black dots in the reflection image inverted, contrast adjusted, and put in the red channel.
click here for reflection microscopy used in collagen matrix