Zeiss LSM 700 - RB 2 601
Cell Biology
To use microscope you must be affiliated with the Department and trained by Michael Cammer or Yan Deng. Please contact Dr. Don Ryoo if you have any questions about eligibility.
Sign up in advance to use microscope required:
https://nyulangonesignups.resourcescheduler.net/resourcescheduler/schedule.asp?Topic=RES&TopicId=81
All files must be saved on Drive D:
NO USER ICONS ONTHE DESKTOP.
Files stored on Drive C: may be deleted!
All files should be copied to Medical Center maintained server.
If you are asked to install an update, just say no!
About the microscope
| Laser lines |
Useful for... |
| 405 nm
|
Dapi, CFP, Pacific Blue |
| 488 nm |
FITC, Alexa488, GFP, YFP |
| 555 nm |
RFP, mCherry, rhodamine, Cy3, Alexa568, Alexa594, propidium iodide |
| 639 nm |
Cy5, Alexa647, To-Pro |
You may image two probes at a time.
Please ask for help checking that you don't have spillover from one channel into another. Sequential scan methods can prevent this. The Smart Setup settings are not always smart and please see the warning in the Quickstart Guide below about using methods from the pull down menu.
Lenses:
- 10X plan neofluar N.A. 0.17 air
- 20X plan apochromat N.A. 0.75 air
- 40X C apochromat N.A. 1.2 water (example image full size)
- 63X plan apochromat 1.4 oil
- 100X plan apochromat N.A. 1.4 oil (recommend do not use this lens; the 63X lens is brighter and las longer working distance)
If you want the highest resolution images, the 40X N.A. 1.2 and the 63X N.A. 1.4 are the best lenses.
Quickstart Guide - The scope has moved a few times so the precise placement in the photo is not correct, but the basic steps are correct.

Please leave the Hg power supply under the table at an angle to the outside right.
When is is squared off facing forward under the table, 1.) people kick the switch and 2.) it is harder to reach the power switch. |
1. Hg arc lamp for viewing fluorescence by eye.
2. Power strip switch.
3. Other power strip switch (order does not matter).
4. Key for lasers (often this is not necessary).
5. Button for computer.
6. When you use settings from the pull down menu, they may have been corrupted by previous users, therefore:
Best way to get settings correct:
Open an image from an earlier session that worked well.
Click on Acquisition tab; this may not work in Locate menu.
Click "Reuse" button to load settings.
Shutdown:
Copy your files over network (preferably to shared server space) or to USB device to take with you.
Check whether someone is coming in soon after you.
Quit software.
Shut down computer.
Switches in reverse order.
|
Log in to LSM User account.
Run Zen software (icon is above center of screen).
DO NOT TOUCH THE MICROSCOPE WHILE THE SOFTWARE IS LOADING.
There are two tabs in the upper left of the software labeled “Locate” and “Acquisition”.
The Locate tab is for looking through the microscope oculars.
The Acquisition tab is for using the confocal microscope.
The Processing tab is for working on images after they have been collected and saved.
Do not use the Maintain tab.
To look by eye, the slider on the right side of the microscope must be pushed in, all the way to the left.
To use the confocal, the slider must be pulled out all the way to the right.

Put slide on the stage.

Put it in the recessed are of the stage closest to you and push it back under the metal tabs.
This means that samples should be at the center of the slide, not at the far edge.
Use joystick to move stage. Button on top toggles from fine to coarse and back.
Find sample. Then set up tracks in Acquisition tab.
Save all images on drive D: only. Files stored on drive C: may be deleted without warning.
All files should be copied to Medical Center maintained server.
Field of View
What you see through the eyepieces covers a much larger area than that scanned by the confocal.
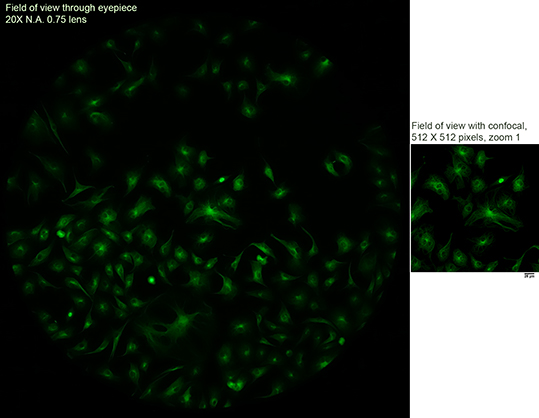
On the left is a snapshot of widefield fluorescence through the eyepiece. On the right is a confocal view at 512 X 512 pixels. (Full size images here.)
But this system has a tiling feature so that you may scan large areas at high resolution and zoom out to get an overview and zom in to get high resolution imaging.
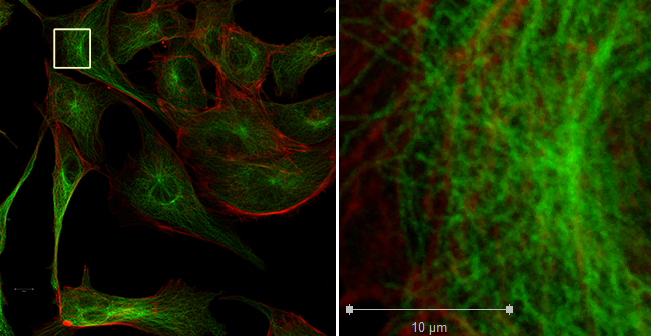
At the left is a mosaic of 3 X 3 tiles using the 100X N.A. 1.4 lens. At the right is the area in the small white box shown at full 1:1 resolution; one pixel in the original image is shown here as one pixel.
On disk, the full size image is 27.7 MB.
General Confocal Best practices:
- The pinhole is what makes the confocal a confocal. Always set at 1AU or down to 0.5 (which means 1 Airy unit) and click the 1AU button each time you change lenses. Explanation why.
If you are opening more than 1.5 Airy units, you should go use a widefield fluorescence scope instead.
Except in exceptional live cell imaging where you understand that you are not being confocal, this is NOT AN ACCEPTABLE WAY TO MAKE IMAGES BRIGHTER. You won't hurt the instrument, but when you write your methods, you won't be accurately describing your microscopy as "confocal".
- Offset. Always use at 0 or 1.
Other numbers are wrong. It is not permissible to removed "background" by setting less than zero.
- Digital gain. The preset is 1. Leave it there.
- Be careful to not saturate your images. For each channel, use the Range Indicator button to make sure pixels are not saturated.
- Example of scan speed, averaging, filtering and noise click here.
- Save files as .czi
Both Imaris and ImageJ open CZI files.
Service information:
LSM 700 Ser Nr. 2601001219
<-- Back
comments, questions, suggestions for this web page: Michael.Cammer@med.nyu.edu or mcammer@gmail.com





